March 3rd, 2026 | 07:40 CET
Paradigm shift in oncological dermatology: Vidac Pharma as an innovator, what are Almirall and Biofrontera doing?
Oncological dermatology is on the cusp of a revolution that could fundamentally change our understanding of cancer treatment. Persistence Market Research estimates that the global market for the treatment of actinic keratosis will reach a volume of around USD 7 billion by 2026. Analysts expect the market to grow to between USD 11.1 and 14.45 billion by the mid-2030s. This momentum is driven by the aging global population, cumulative UV exposure due to changing leisure habits, and increased awareness of the dangers of skin cancer. Currently, established industry giants such as Almirall and Biofrontera dominate the standard of care for actinic keratosis with their proven products. However, Vidac Pharma marks the transition from merely combating symptoms to a revolutionary procedure in oncological dermatology. This makes the company an opportunity for growth-oriented investors seeking access to a completely new class of drugs in the field of metabolic oncology.
time to read: 3 minutes
|
Author:
Nico Popp
ISIN:
VIDAC PHARMA HOLDING PLC | GB00BM9XQ619 , ALMIRALL S.A. EO -_12 | ES0157097017 , BIOFRONTERA AG NA O.N. | DE0006046113
Table of contents:
Author
Nico Popp
At home in Southern Germany, the passionate stock exchange expert has been accompanying the capital markets for about twenty years. With a soft spot for smaller companies, he is constantly on the lookout for exciting investment stories.
Tag cloud
Shares cloud
How the market leader Almirall operates
Almirall's business model is based on a clear focus on medical dermatology, giving the company a leading role in Europe. In fiscal year 2025, Almirall recorded net sales growth of 12.4% to around EUR 1.1 billion, while EBITDA rose by 20.9% to EUR 232.9 million. A key pillar of this success is the marketing of Klisyri for the field treatment of actinic keratosis, which achieved sales growth of 33.9% to EUR 32.8 million. Although this microtubule inhibitor offers patients an ultra-short treatment duration of only five days, the active ingredient primarily interferes with cell division and addresses the problem symptomatically rather than causally. To secure future growth and defend its market position, the group is consistently investing in research and development. In 2025 alone, EUR 138.1 million was spent on research and development.
Biofrontera: Specialized approaches and new mechanisms of action
Biofrontera, on the other hand, occupies a specialized niche in the treatment of actinic keratosis by combining light therapy and pharmacological effects. Its core product, Ameluz, is used in conjunction with the RhodoLED lamp series and is currently benefiting from a new sales strategy and targeted price adjustments in the important US market. For the full year 2025, the company expects revenue of between USD 41.5 and 42.0 million, representing a solid increase of 11% to 13%. The company is also aggressively expanding into new areas and announced positive results from a Phase 3 study for the treatment of the extremities, neck, and trunk in February 2026. Despite its high efficacy and expansion into new areas of application, however, Biofrontera's preferred combination therapy remains costly and often painful for patients.
The revolutionary: What makes Vidac Pharma different
In contrast to established therapies, Vidac Pharma targets a fundamental metabolic hallmark of cancer: the so-called Warburg effect. Cancer cells preferentially generate energy through aerobic glycolysis, a process strongly associated with overexpression of the enzyme hexokinase-2 (HK2). **Malignant cancer cells produce far too much of this enzyme. It then docks at the wrong place on the cell's "power plants," which blocks the diseased cell's natural self-destruction program. This allows the tumor to continue growing unchecked. Vidac's leading drug candidate, VDA-1102, breaks this unnatural bond. This new mechanism reactivates the cancer cell's natural distress signals, causing it to die in a targeted manner. In addition, the cell's metabolism is reprogrammed to normal, healthy energy production.
Clinical validation and technological scalability
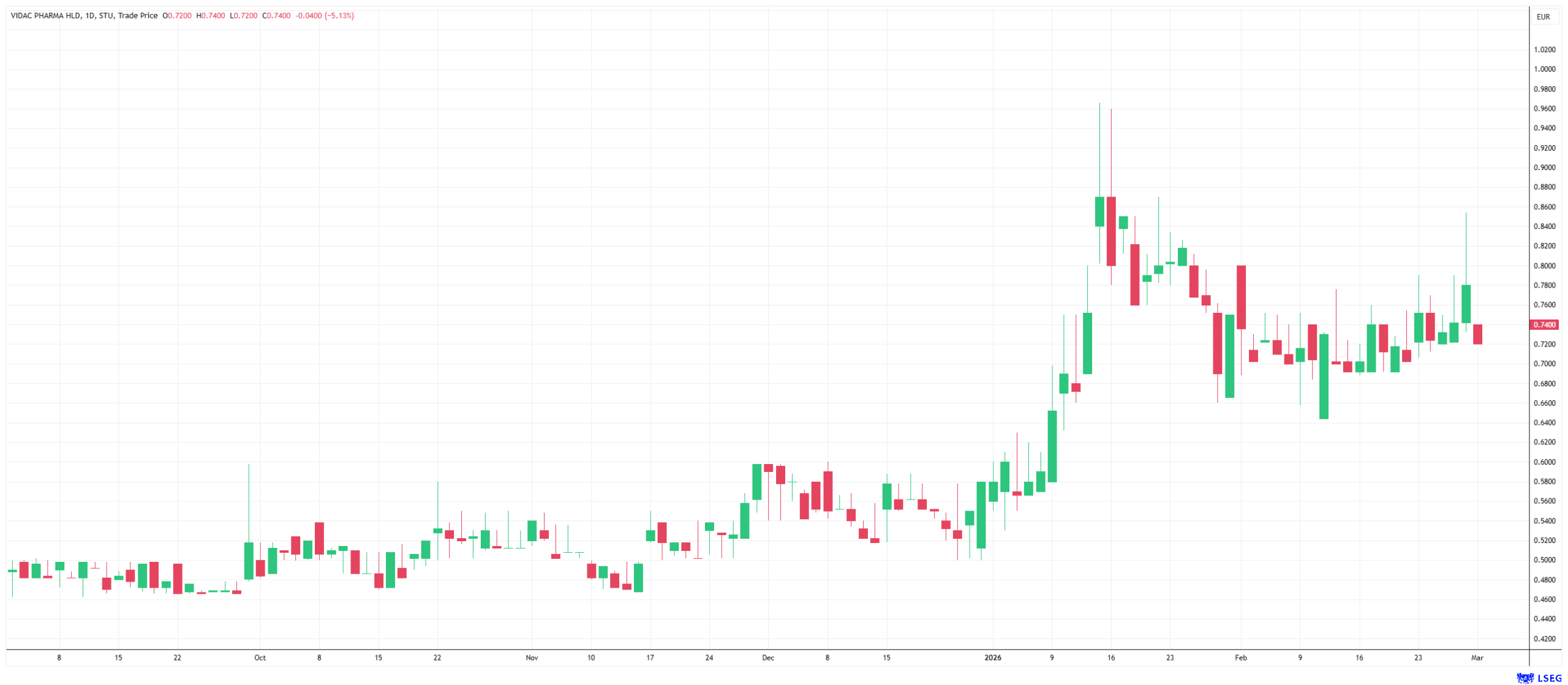
Vidac is currently verifying the theoretical superiority of this approach through extensive patient studies. At the end of February, the company officially began dosing the first patient in a Phase 2b clinical trial for VDA-1102 in high-risk patients with actinic keratosis. This study, which is being conducted at the CentroDerm specialist center in Wuppertal under the direction of Prof. Thomas Dirschka, deliberately focuses on highly proliferative lesions with a high risk of direct transition to invasive carcinoma. In a Phase 2a study, the compound achieved a statistically significant 50% reduction in lesion count, while demonstrating good tolerability and minimal local skin reactions. A drug that effectively heals without compromising quality of life during treatment due to pain or redness has the potential to revolutionize the entire market for field therapies.
Vidac Pharma's innovative strength goes far beyond existing oncological approaches, as evidenced by the simultaneous expansion of its technology platform. Also in February, the company expanded its commitment by launching an in vivo preclinical program for psoriasis, as scientific studies have shown that the enzyme hexokinase-2 is also significantly elevated in psoriatic lesions (symptoms of psoriasis). This unique technological standing positions Vidac Pharma as an agile first mover in industry. Independent analysts at Sphene Capital reaffirm their "Buy" recommendation with a price target of EUR 4.10 and emphasize that the market has not yet adequately priced in the far-reaching significance of cellular correction. This opens up direct access for investors to a pioneer in metabolic cancer medicine that aims to permanently shift the boundaries of conventional dermatology.
Conflict of interest
Pursuant to §85 of the German Securities Trading Act (WpHG), we point out that Apaton Finance GmbH as well as partners, authors or employees of Apaton Finance GmbH (hereinafter referred to as "Relevant Persons") may hold shares or other financial instruments of the aforementioned companies in the future or may bet on rising or falling prices and thus a conflict of interest may arise in the future. The Relevant Persons reserve the right to buy or sell shares or other financial instruments of the Company at any time (hereinafter each a "Transaction"). Transactions may, under certain circumstances, influence the respective price of the shares or other financial instruments of the Company.
In addition, Apaton Finance GmbH is active in the context of the preparation and publication of the reporting in paid contractual relationships.
For this reason, there is a concrete conflict of interest.
The above information on existing conflicts of interest applies to all types and forms of publication used by Apaton Finance GmbH for publications on companies.
Risk notice
Apaton Finance GmbH offers editors, agencies and companies the opportunity to publish commentaries, interviews, summaries, news and the like on news.financial. These contents are exclusively for the information of the readers and do not represent any call to action or recommendations, neither explicitly nor implicitly they are to be understood as an assurance of possible price developments. The contents do not replace individual expert investment advice and do not constitute an offer to sell the discussed share(s) or other financial instruments, nor an invitation to buy or sell such.
The content is expressly not a financial analysis, but a journalistic or advertising text. Readers or users who make investment decisions or carry out transactions on the basis of the information provided here do so entirely at their own risk. No contractual relationship is established between Apaton Finance GmbH and its readers or the users of its offers, as our information only refers to the company and not to the investment decision of the reader or user.
The acquisition of financial instruments involves high risks, which can lead to the total loss of the invested capital. The information published by Apaton Finance GmbH and its authors is based on careful research. Nevertheless, no liability is assumed for financial losses or a content-related guarantee for the topicality, correctness, appropriateness and completeness of the content provided here. Please also note our Terms of use.